-1693225095.png)
Masterbatch for medical products
.png)
Plastic, the medical marvel
Cutting-edge medicine without plastics – would be impossible today. It is the outstanding and flexible properties of plastics in particular that make this material so attractive and important for medical technology.
Unlike glass, metal or ceramics, plastic scores with its ease of processing, fracture resistance, low weight and the possibility of designing it for specific requirement profiles.
Our Maxithen® MED series consists of a range of colour masterbatch for almost all production technologies as well as a wide range of applications, including the colouring of needle holders.
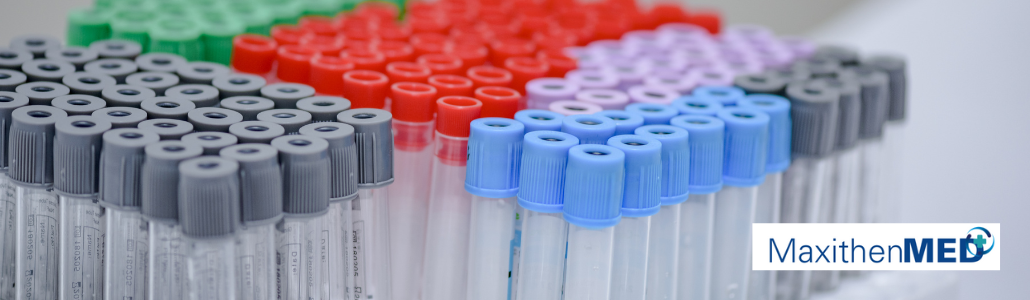
Needle holder according to ISO 6009
With our Maxithen® MED series, we offer a range of colour masterbatch for the colouring of needle holders. The colours comply with the international ISO 6009 standard that aims to enable the quick visual recognition of needle diameters for injections. The colours used for the identification of the needles are available as transparent or covering colours. The colour code correlates with normal, thin and extra-thin wall thicknesses of the needle.
-1693225408.png)
-1695983091.png)
Masterbatches designed for ISO 10993
Gabriel-Chemie Group offers a range of masterbatches which is designed to meet the requirements of ISO 10993. This product range consists in PE and PP based carrier masterbatches as well as laser marking colour-additive masterbatches.
The product range helps reduce product development time due to being ready-made products. We support our customers through a solid technical support and, of course, through an extensive product documentation. The product range was not tested on animals in an attempt to avoid animal pain.
-
Press Release ISO 10993